|
The electron configuration of the given elements are as following: Nitrogen is 1s22s2. Elements in other groups vary in their reactivity but are generally less reactive than elements in groups \(1,\,2,\,16,\) or \(17.\) Valence Electrons and Electricity 1. These include Nitrogen, Phosphorous, Arsenic, Antimony and Bismuth. These elements already have a full outer energy level, so they are very stable. 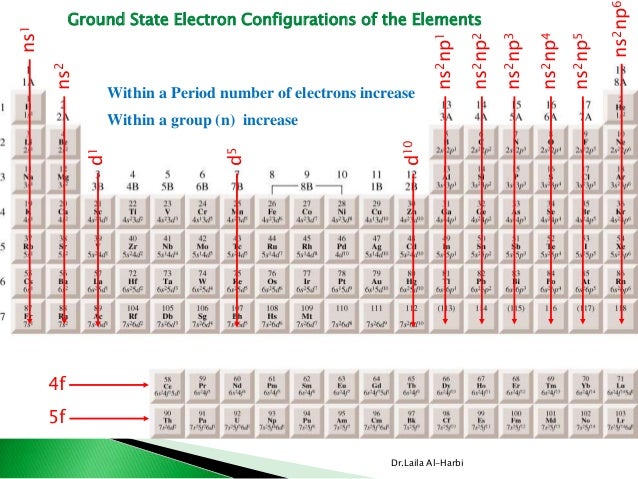 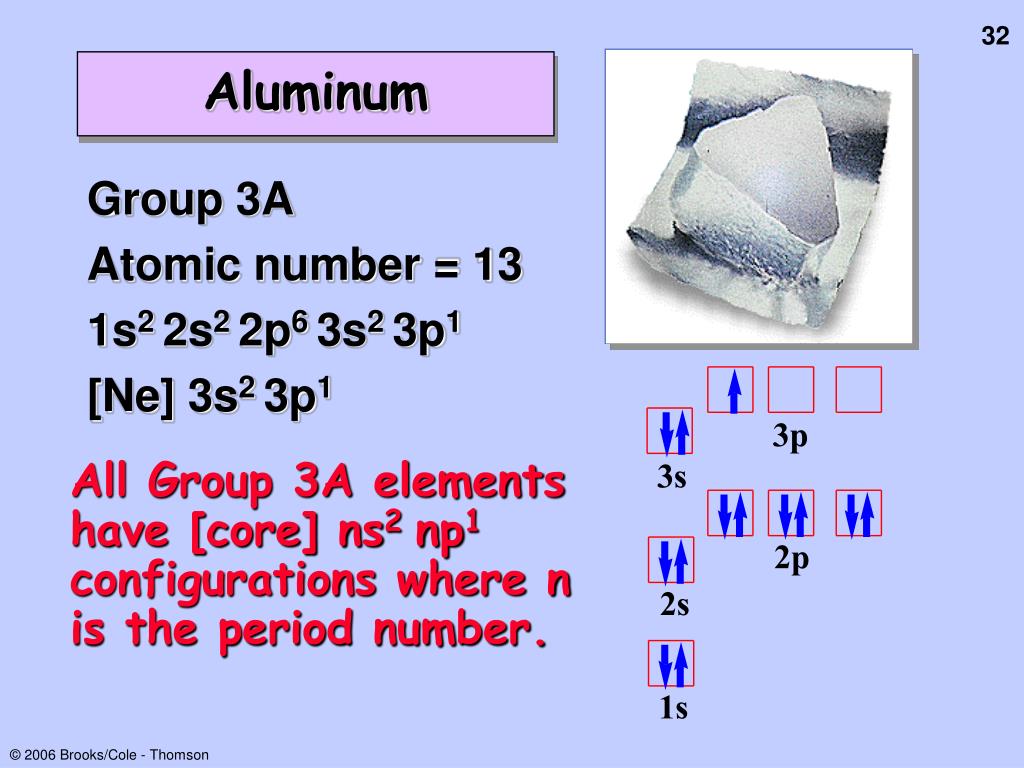
filled valence shell electron configuration, ns2 np3 where as fluorine has. In the long form of the periodic table, the valence shell electronic configuration of 5s☥p4 corresponds to the element present in: Group 16 and period 6 Group 17 and period 6 Group 16 and period 5 Group 17 and period 5 Answer: Group 16 and period 5. The best explanation is that : (A) the valency electrons in F are on the aver. Atoms of group 18 elements have eight outer-shell electrons (or two in the case of helium). The number of elements in the 5th period of the periodic table is. These subatomic particles carry a negative charge of \(1.602 \times \right),\) it readily accepts the electron given by the sodium atom. In an electron configuration under the general form of ns1, the n represents the period number and the 1 represents the group number, and therefore, the element would be in group 1. Valence Electrons: Concepts to UnderstandĮlectrons are tiny subatomic particles that revolve around the nucleus in energy shells called orbits.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
